This post was originally published on here
Newswise — Biomethanation provides an energy-efficient, low-carbon alternative to thermochemical gas conversion, turning biomass-derived syngas into biomethane for circular energy systems. The performance of this process depends on balanced microbial metabolism, where hydrogenotrophic methanogens reduce CO₂ using H₂, supported by acetogens and syntrophic partners. However, syngas composition fluctuates during industrial operation, and the metabolic response to hydrogen excess is poorly understood. Traditional studies observed performance drops at high H₂ supply, but lacked molecular-level mechanistic explanation regarding microbial regulation and viral interactions. Due to these uncertainties, deeper investigation into microbial and viral responses under hydrogen-rich conditions is needed.
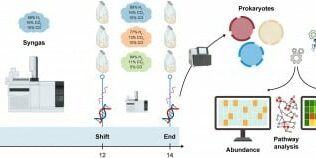
Researchers from the University of Padua reported on a 2025 early-access study (DOI: 10.1016/j.ese.2025.100637) in Environmental Science and Ecotechnology demonstrating how hydrogen surplus alters microbiome metabolism and triggers viral defense responses in syngas-converting systems. Using genome-resolved metagenomics, metatranscriptomics and virome profiling, the team monitored microbiomes as syngas composition shifted from optimal ratios to hydrogen-rich conditions. Their findings uncover a stress-driven metabolic reorganization and highlight phage dynamics as a significant ecological dimension in biomethanation efficiency.
The study cultivated thermophilic anaerobic microbiomes under three syngas compositions and applied multi-omics analysis to track responses before and after hydrogen increase. Under near-optimal gas ratios, methane yield improved and the dominant methanogen Methanothermobacter thermautotrophicus maintained stable gene expression. However, when hydrogen supply exceeded stoichiometric demand, methane production declined and transcriptome analysis revealed strong metabolic repression. Key methanogenesis genes—including mcr, hdr, mvh, and enzymes in CO₂-to-CH₄ reduction—were significantly downregulated.
Simultaneously, M. thermautotrophicus activated antiviral defense systems, upregulating CRISPR-Cas, restriction-modification genes, and stress markers such as ftsZ. Virome mapping identified 190 viral species, including phages linked to major methanogens and acetogens. Some viruses showed reduced activity, suggesting defense-driven suppression, while others exhibited active replication patterns. In contrast, several acetogenic taxa—including Tepidanaerobacteraceae—enhanced expression of Wood–Ljungdahl pathway genes (cdh, acs, cooF, cooS) to boost CO/CO₂ fixation and act as electron sinks. This reprogramming indicates a shift from methanogenesis to carbon-fixation-dominant metabolism when hydrogen is excessive.
The authors emphasize that hydrogen excess creates a regulatory bottleneck, pushing methanogens into stress mode while enabling acetogens to take over carbon metabolism. They note that viral interactions—previously overlooked in biomethanation—play a major role in shaping community stability. The team points out that CRISPR-Cas activation and phage suppression indicate a defensive state, suggesting that virome dynamics must be considered in bioreactor design.
This research provides molecular-level evidence that hydrogen oversupply can destabilize methane production, highlighting the need for gas-ratio control in industrial reactors. Understanding how microbial populations reprogram under stress can guide engineering of more resilient biomethanation systems, enabling consistent biomethane yields even with variable feedstocks. The insights into phage-microbe interactions further suggest potential for virome-aware reactor management strategies, including microbial community design, phage monitoring, or antiviral interventions. These findings support future development of carbon-neutral gas-to-energy technologies and scalable waste-to-resource platforms.
###
References
DOI
Original Source URL
https://doi.org/10.1016/j.ese.2025.100637
Funding information
This work was supported by the LIFE20 CCM/GR/001642 – LIFE CO2toCH4 of the European Union LIFE + program and the European Union’s Horizon 2020 research and innovation program under grant agreement No 101084405 (CRONUS).
About Environmental Science and Ecotechnology
Environmental Science and Ecotechnology (ISSN 2666-4984) is an international, peer-reviewed, and open-access journal published by Elsevier. The journal publishes significant views and research across the full spectrum of ecology and environmental sciences, such as climate change, sustainability, biodiversity conservation, environment & health, green catalysis/processing for pollution control, and AI-driven environmental engineering. The latest impact factor of ESE is 14.3, according to the Journal Citation ReportsTM 2024.